 
The Rydberg formula remained unexplained until the first successful model of the hydrogen atom was proposed in 1913. Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. 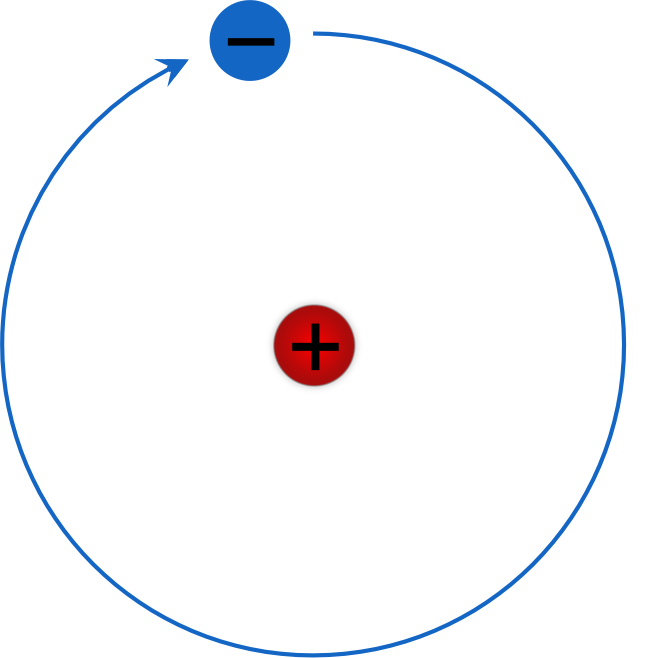
The Rydberg formula for hydrogen gives the exact positions of the spectral lines as they are observed in a laboratory however, at the beginning of the twentieth century, nobody could explain why it worked so well. 
As you may guess, there are infinitely many such spectral bands in the spectrum of hydrogen because \(n_f\) can be any positive integer number. When \(n_f = 6\), we have the Humphreys series. When \(n_f = 5\), the series is called the Pfund series. When \(n_f = 4\), the series is called the Brackett series. When \(n_f = 3\), the series is called the Paschen series. Understanding Bohr's model requires some knowledge of electromagnetic radiation (or light).īohr's key idea in his model of the atom is that electrons occupy definite orbitals that require the electron to have a specific amount of energy.). In 1913, the Danish physicist Niels Bohr proposed a model of the electron cloud of an atom in which electrons orbit the nucleus and were able to produce atomic spectra. U(r) ke2 r, where k 1 / 40 and r is the distance between the electron and the proton. Figure 2 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. In the electric field of the proton, the potential energy of the electron is. Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. With the assumption of a fixed proton, we focus on the motion of the electron. These difficulties cast a shadow on the planetary model and indicated that, eventually, it would have to be replaced. Figure 8.2.1: A representation of the Bohr model of the hydrogen atom. 1: The energy levels of the electrons can be viewed as rungs on a ladder. My objectives were: (1) To present Bohr model as a semi-classical approach (temporary solution) to atomic physics. Furthermore, Rutherford's model was unable to describe how electrons give off light forming each element's unique atomic spectrum. One of the weaknesses of Bohrs model was that he could not offer a reason why only certain energy levels or orbits were allowed. This lecture was presented to undergraduate students at San Jose State University. If the electron circling the nucleus in an atom loses energy, it would necessarily have to move closer to the nucleus as it loses energy, and would eventually crash into the nucleus. This is, after all, how we produce TV signals. The Bohr radius ( ) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. It was already known that when a charged particle (such as an electron) moves in a curved path, it gives off some form of light and loses energy in doing so. Unfortunately, there was a serious flaw in the planetary model. 
The model has a special place in the history of physics because it introduced an early quantum theory, which brought about new developments in scientific thought and later culminated in. \): Niels Bohr with Albert Einstein at Paul Ehrenfest's home in Leiden (December 1925). Historically, Bohr’s model of the hydrogen atom is the very first model of atomic structure that correctly explained the radiation spectra of atomic hydrogen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
